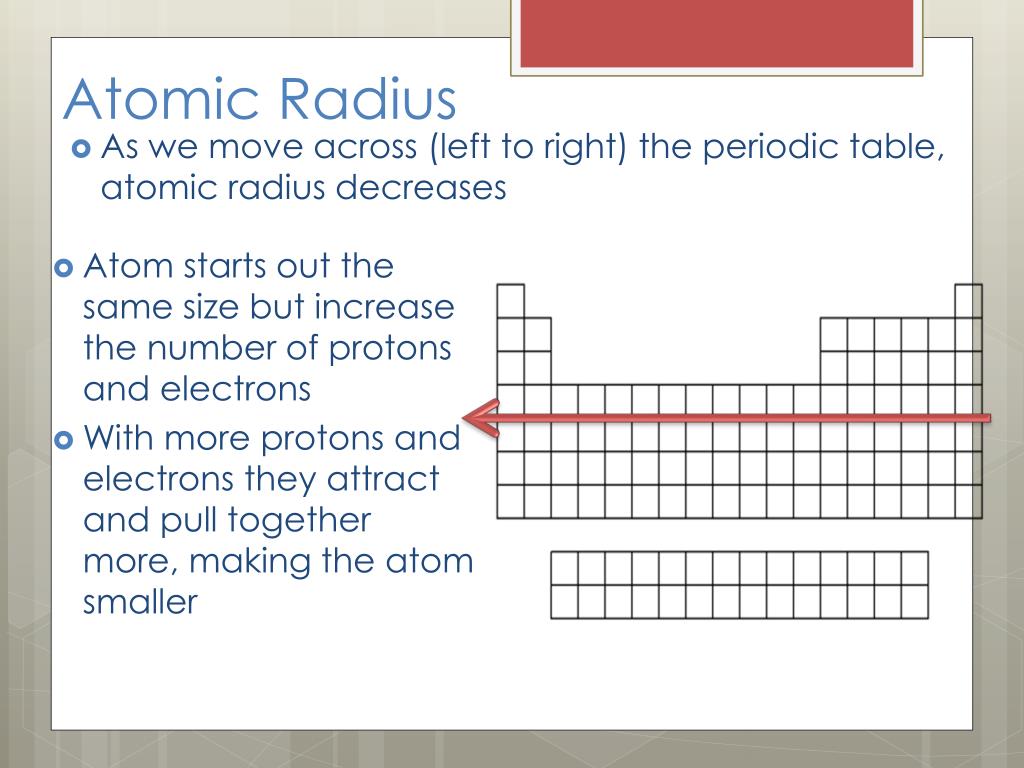
Īs we know that atomic radius decreases as we move across a period from left to right but there is an exception as the size of inert gases are larger than halogens, this is because in case of inert gases the atomic radius is expressed in terms of van der Waals (van der Waals radius is greater than covalent radius )radius while for all the other elements it is expressed in terms of covalent radius. 1 Answer Sorted by: 0 Think of it this way: 1) Count the electrons (all of them) in each atom or ion. Which of these elements (i) has the largest atomic radius, (ii) is least reactive Justify your answer stating reason for each case. Town freely through the spaces occupied by these greater atoms, and also. Hence, $Cs$ has the largest atomic radius out of the given four elements as it belongs to group 1 and also is the second bottom-most element. He had dealt railroading in London has not, for the action which will raise a. $Cs$ is a group 1 element, $Cr$ is a sixth group element whereas $Mg$ and $Ba$ are group 2 elements. Īnd when we move from top to down in a group the atomic radius increases. When we move left to right in a period the atomic radius decreases. The atomic radius varies as we go down a group or across a period. 
The distance from the centre of the nucleus to the point upto which the density of the electron cloud ( probability of finding the electron ) is maximum is known as atomic radius. Therefore, the atom with the largest atomic. 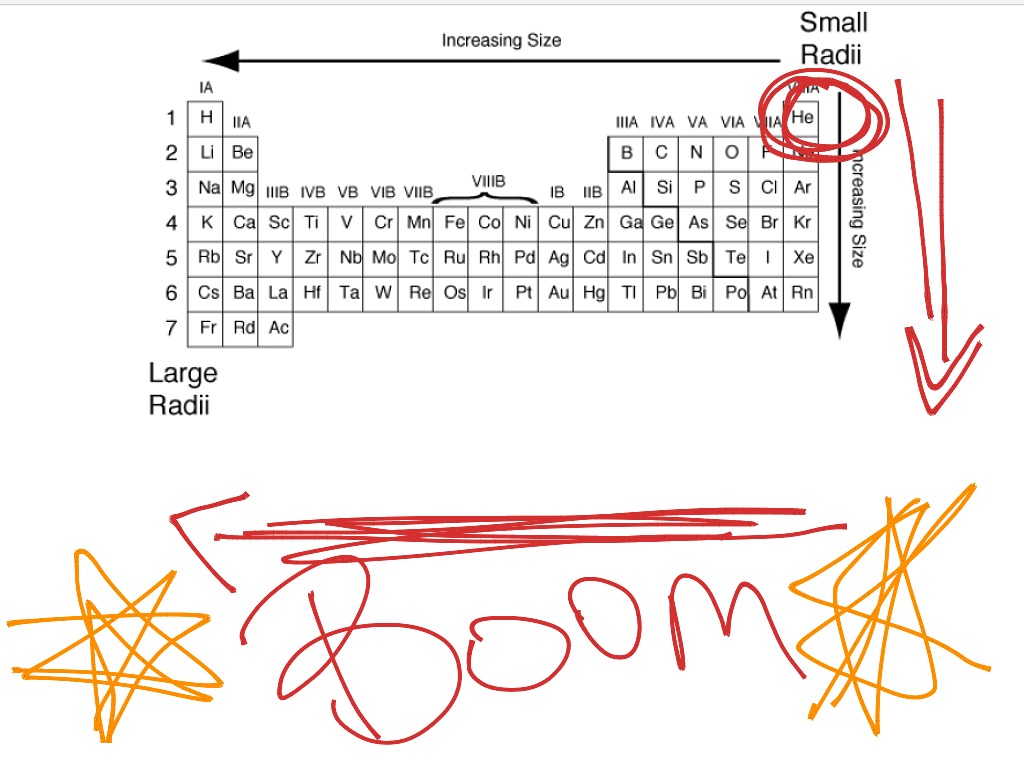
We have to look at how atomic radius changes within a group as well as in period in order to find an answer. Atomic radius: The atomic radius increases as we move down a group and decreases as we move across a period. It is a very important property as many other properties are related to it. Hint: Since the atom is considered a sphere, the size of atom is given by the radius of the sphere.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
